Health Care Roundup: More Drug Pricing News; Clinical Trial Challenges; Trump Promises Action on Surprise Billing; Fake Cancer Cures; More
Subscribe to our email list and receive these updates in your email box each week »
HEALTH CARE HIGHLIGHTS
Drug Pricing, Drug Pricing, and More Drug Pricing!
This week, the House and Senate held several hearings related to health care, including a House Ways and Means Committee hearing titled “Protecting Americans with Pre-Existing Conditions.” The hearing illustrated the stark difference between the two parties’ health care goals.
There were also dueling drug pricing hearings this week, one from the House Committee on Oversight and Reform titled, “Examining the Actions of Drug Companies Raising Prescription Drug Prices.” The Senate Finance Committee also held a hearing titled, “Drug Pricing in America: A Prescription for Change, Part I.”
Robert Pear of the New York Times provided an overview of the hearings:
On Thursday, Health and Human Services (HHS) Secretary Alex Azar announced a policy proposal that would eliminate rebates from drug manufacturers to insurers and pharmacy benefit managers in the Medicare program, which the department claims would lower the out-of-pocket spending on prescription drugs at the pharmacy. Azar then called on Congress to pass the proposal into law banning rebates and extending it to the commercial market. Under draft rules, drug manufacturers would be allowed to offer rebates directly to consumers but would no longer be able to give rebates to pharmacy benefit managers. Stay tuned as NCCS analyzes this proposal and its potential impacts on cancer survivors.
NCCS recently commented on another drug pricing proposal by HHS. This proposed rule would change Medicare Part D’s protected classes drug coverage. In response, NCCS joined with other patient and provider groups of the Cancer Leadership Council (CLC) to comment on how these proposed changes could impact patients. The proposed rule would give Medicare plans the option to limit coverage of drugs in six categories, including cancer drugs. In the comment letter, the groups urge HHS to reconsider these changes as they would create barriers to treatments that cancer patients need. One major reservation articulated in the letter concerned the impact of the broader use of prior authorization and step therapy for drugs in protected classes. Access to only one drug in a specific drug class would undermine the ability to receive appropriate and recommended treatment.
CHART OF THE WEEK
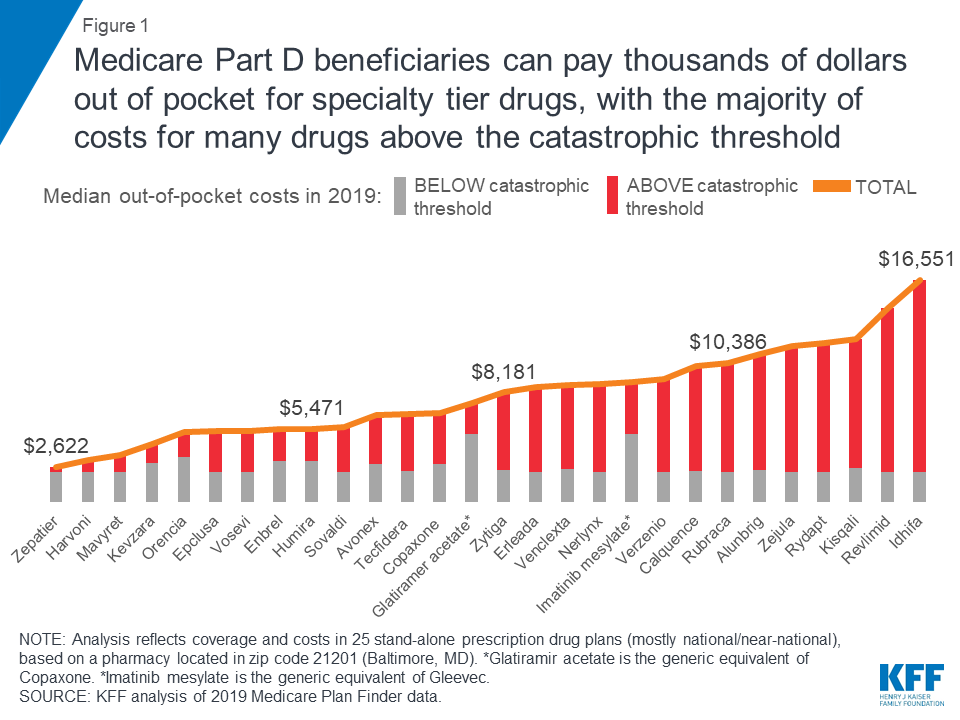
The Out-of-Pocket Cost Burden for Specialty Drugs in Medicare Part D in 2019
Kaiser Family Foundation
A recent analysis from Kaiser Family Foundation found that despite Medicare’s protections, Part D enrollees with serious health problems can face thousands of dollars in annual out-of-pocket spending.
IMPORTANT READS
Clinical Trial Exclusions: Are Researchers Missing Out?
Cure Magazine
Expanding access and increasing recruitment for clinical trials is an ongoing issue for NCCS. This article discusses the many challenges of recruiting patients to participate in clinical trials investigating immunotherapy drugs.
Mohammad Jahanzeb, M.D., a professor of hematology/oncology at the University of Miami Sylvester Comprehensive Cancer Center, says:
Trump boosts fight against surprise medical bills
The Hill
Momentum is building for action to prevent patients from receiving massive unexpected medical bills, aided by President Trump, who is vowing to take on the issue. Calls for action against so-called "surprise" medical bills have been growing, spurred by viral stories like one involving a teacher in Texas last year who received a $108,000 bill from the hospital after his heart attack.
The modern tragedy of fake cancer cures
STAT News
A Jerusalem Post article making the rounds on social media claims that an Israeli biotech company will have “a complete cure for cancer” within a year. Matthew Herper explains why the claim is not true, writing:
Ads for Short-Term Plans Lacking ACA Protections Swamped Consumers’ Online Searches
Kaiser Health News
A new study shows that consumers looking for health insurance online last fall — using search terms such as “Obamacare plans,” “ACA enroll” and “cheap health insurance” — were most often directed to websites that sell short-term limited duration health plans that don’t comply with the ACA’s consumer protections.
Consolidation of health insurance and drug benefits is back
Axios
Through rapid consolidation, five companies – Anthem, Cigna, CVS Health, Humana and UnitedHealth – will control insurance and pharmacy benefits for more than 125 million Americans.
Related Posts
https://canceradvocacy.org/blog/health-care-roundup-jan-25-2019/